Ольга Колчина
ISO 22716 – международный стандарт, представляющий руководящие указания по Надлежащей Производственной Практике (GMP) для парфюмерно-косметической продукции. В большинстве стран мира организации, которые производят или вводят в обращение на рынок парфюмерно-косметическую продукцию, должны декларировать соответствие условий производства требованиям ISO 22716 (GMP).

Международный стандарт ISO 22716 – это
производство косметической продукции в управляемых условиях | условие для старта контрактного производства и выхода на международное сотрудничество, рынки |
системный подход к созданию безопасного продукта | выполнение законодательных требований большинства стран мира |
прослеживаемость в ходе всей производственной цепочки | доверие и удовлетворенность потребителей |
система вовлечения, обучения и развития сознательного отношения персонала к производству | получение уверенности перед проверками государственных надзорных органов благодаря планомерной работе по выявлению, внедрению и контролю соблюдения законодательных и прочих нормативных требований |
входной контроль, межоперационный контроль и контроль готовой продукции для уверенности в соответствии продукта требованиям качества и безопасности | улучшение имиджа компании |
Требования ISO 22716 охватывают регулирование следующих областей:
- персонал
В этой области вводятся подходы к управлению квалификацией, обучением, здоровьем и гигиеной персонала парфюмерно-косметического производства.
- инфраструктура
Значимая для ISO 22716 инфраструктура это- здания и сооружения, производственное оборудование, средства мониторинга и измерений, вентиляция, водоподготовка. GMP вводит требования к качеству и безопасности инфраструктуры, ее поддержанию в исправности и готовности к чрезвычайным ситуациям для непрерывного обеспечения бизнеса.
- правила системы Надлежащей Производственной Практики
Система управления согласно ISO 22716 реализуется через следование регламентам (часто стандартным операционным процедурам SOP) и ведение подтверждающих записей о соответствии выполненных процессов.
Внедрение требований ISO 22716 охватывает следующие этапы:
- диагностический аудит;
- обучение персонала требованиям GMP;
- разработка процедур, их документирование;
- внедрение разработанных подходов в ежедневную практику;
- оценка и улучшение системы Надлежащей Производственной Практики.
Сертификация ISO 22716
Сертификация ISO 22716 выполняется по неаккредитованной процедуре. Сертификация не заменяет обязательных проверок регулирующих органов стран, где вводится в обращение парфюмерно-косметическая продукция. Целью сертификации является проверка и подтверждение способности организаций управлять процессами для производства безопасной продукции, что подтверждается итоговым документом, сертификатом GMP известной международной экспертной организацией.
Как получить сертификат ISO 22716?
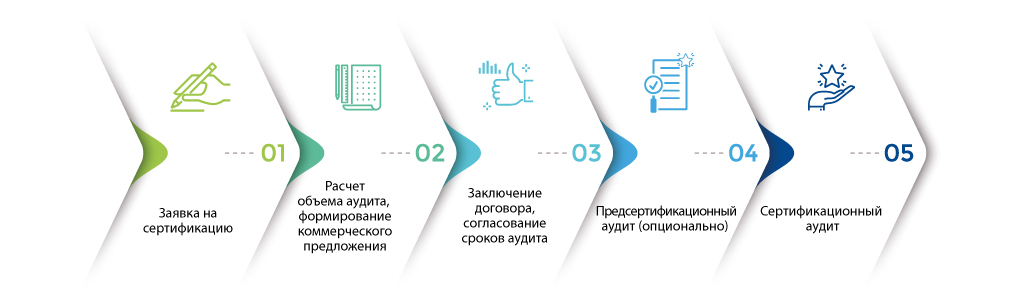
Кроме внешних преимуществ (сертификат ISO 22716 предъявляется заинтересованным сторонам) сертификационный аудит является инструментом обмена актуальными знаниями, опытом, практиками и помогает улучшению организации.
Мы будем рады
- получить от вас заявку на сертификацию;
- представить услуги по аудиту поставщиков для производителя парфюмерно-косметической продукции;
- пригласить вас на наши обучающие мероприятия;
- рассказать вам о нашем опыте.
Департамент комплексных проектов

